Cancer is a complex and often challenging disease that affects millions of people worldwide. In recent years, there has been a growing interest in the role of genetics in understanding and treating cancer. This fascinating and rapidly evolving field of research offers new insights into the genetic basis of cancer, as well as potential opportunities for personalized treatments. In this blog post, we will delve into the various ways in which genetics contributes to our understanding of cancer and the development of innovative treatment strategies. From the basics of genetic mutations and inherited factors to the cutting-edge techniques of genomics and gene editing, we will explore the key concepts and breakthroughs that are shaping the future of cancer care. Join us as we unravel the intricate relationship between genetics and cancer, and discover the potential impact of these advancements on the way we approach this challenging disease.
Table of Contents
Introduction to Genetics and Cancer
Genetics play a crucial role in the development and progression of cancer. Understanding the genetic factors that contribute to cancer can provide valuable insights into its prevention and treatment.
Genetic mutations can increase the risk of cancer. These mutations may be inherited or acquired over time due to environmental factors. Certain inherited genetic factors have been identified as predisposing individuals to certain types of cancer.
Advancements in genomics have revolutionized the way we understand and approach cancer. By unlocking the cancer genome, researchers and healthcare professionals are able to identify specific genetic abnormalities that drive cancer development.
As we delve deeper into the world of genetics and cancer, the potential for targeted therapy and personalized cancer treatments becomes more apparent. This has paved the way for gene editing technologies that hold promise for revolutionizing cancer treatment in the future.
Genetic Mutations and Cancer Risk
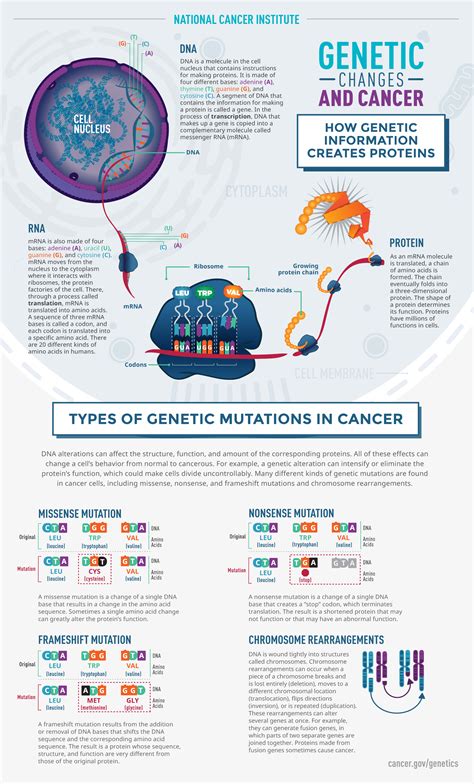
Genetic mutations are alterations in the DNA sequence that can increase the risk of developing cancer. These mutations can be inherited, occurring in the germline, or acquired, arising during a person’s lifetime due to environmental factors or errors in DNA replication.
Individuals with germline mutations in certain genes, such as BRCA1 and BRCA2, have a higher risk of developing breast and ovarian cancer. On the other hand, acquired mutations, such as those in the TP53 gene, can lead to an increased risk of various types of cancer, including breast, colorectal, and lung cancer.
Understanding the relationship between genetic mutations and cancer risk is crucial for cancer prevention and early detection. Genetic testing can identify individuals with an increased susceptibility to cancer, allowing for personalized screening and preventive measures. Additionally, knowledge of specific mutations can inform targeted treatment options for individuals diagnosed with cancer.
Overall, genetic mutations play a significant role in determining an individual’s susceptibility to cancer, highlighting the importance of genetic testing and personalized cancer care in reducing the burden of this disease.
Inherited Genetic Factors and Cancer
Genetic factors can play a significant role in an individual’s risk for developing cancer. Inherited genetic mutations can be passed down from generation to generation, increasing the likelihood of certain types of cancer within a family. These hereditary genetic factors can significantly influence a person’s susceptibility to developing cancer.
Individuals with a family history of certain types of cancer may be at a higher risk due to inherited genetic factors. For example, certain mutations in the BRCA1 and BRCA2 genes are associated with an increased risk of developing breast and ovarian cancer. Understanding these inherited genetic factors is crucial in assessing an individual’s cancer risk and developing personalized screening and prevention strategies.
Furthermore, inherited genetic factors can also impact the effectiveness of cancer treatments. Certain genetic variations can influence how an individual responds to specific cancer therapies, leading to differences in treatment outcomes. By identifying these inherited genetic factors, healthcare providers can personalize cancer treatments to optimize their effectiveness and minimize potential side effects.
Overall, the role of inherited genetic factors in cancer is complex and multifaceted. It underscores the importance of genetic testing and counseling to identify individuals at higher risk and tailor personalized cancer prevention and treatment strategies. By understanding and addressing inherited genetic factors, we can make significant strides in reducing the burden of cancer on individuals and families.
Genomics: Unlocking the Cancer Genome
Genomics is a field of study that focuses on the structure, function, and mapping of an organism’s complete set of DNA, also known as its genome. In the context of cancer, genomics has revolutionized our understanding of the disease by allowing us to delve into the genetic abnormalities that drive cancer development.
Through the use of advanced technologies, scientists have been able to sequence the entire cancer genome, identifying mutations and alterations that contribute to the initiation and progression of cancer. This has paved the way for personalized cancer treatments tailored to an individual’s genetic makeup, leading to more effective and targeted therapies.
Furthermore, genomics has provided valuable insights into the heterogeneity of cancer, revealing the diverse genetic landscapes that exist within tumors. This knowledge has enabled researchers to identify potential drug targets and develop new strategies for combating the disease.
Overall, genomics is unlocking the cancer genome, shedding light on the intricate molecular mechanisms underlying cancer and offering hope for improved diagnostics and treatment options in the future.
The Impact of Tumor Suppressor Genes
Tumor suppressor genes play a crucial role in preventing the development of cancer by regulating cell growth and division. When these genes are mutated, the normal growth-inhibiting mechanisms are disrupted, leading to the uncontrolled proliferation of cells, which is a hallmark of cancer. The impact of tumor suppressor genes on cancer development is significant, as their dysfunction can result in the formation of malignant tumors.
One of the most well-known tumor suppressor genes is the p53 gene, which is often referred to as the guardian of the genome. Mutations in the p53 gene can lead to increased susceptibility to cancer, as it is unable to effectively inhibit the growth of abnormal cells. This highlights the critical role that tumor suppressor genes play in maintaining genomic stability and preventing the formation of cancerous cells.
Furthermore, the impact of tumor suppressor genes extends beyond their role in preventing the initiation of cancer. These genes also play a crucial part in the regulation of DNA repair mechanisms, which are essential for maintaining the integrity of the genome. Loss of function mutations in tumor suppressor genes can compromise the ability of cells to repair damaged DNA, leading to the accumulation of mutations and an increased risk of cancer development.
In conclusion, the impact of tumor suppressor genes on cancer development is profound, as their role in regulating cell growth and maintaining genomic stability is crucial for preventing the initiation and progression of cancer. Understanding the function of these genes and their mechanisms of action is essential for developing targeted therapies that can restore their normal function and prevent the development of cancer.
Oncogenes: Drivers of Cancer Development
Oncogenes play a crucial role in the development of cancer. These genes were originally identified as the culprits that drive the uncontrolled growth of cancer cells. When these oncogenes are activated, they promote cell proliferation, inhibit apoptosis, and enhance the invasion and metastasis of cancer cells.
One of the most well-known oncogenes is the RAS gene. Mutations in the RAS gene can lead to the continuous activation of signaling pathways that drive cell growth and division, ultimately resulting in the formation of tumors. Another important group of oncogenes is the MYC family of genes. These genes regulate cell cycle progression and promote cell proliferation when overexpressed, contributing to the development of cancer.
It is important to note that oncogenes can be activated through various mechanisms, such as point mutations, gene amplification, or chromosomal translocations. Additionally, the activation of oncogenes can be influenced by environmental factors, such as exposure to carcinogens or viral infections.
Understanding the role of oncogenes in cancer development has paved the way for the development of targeted therapies that aim to inhibit the activity of these genes and halt the progression of cancer. By specifically targeting the aberrant function of oncogenes, researchers and clinicians are able to design more effective and personalized treatment strategies for cancer patients.
Pharmacogenomics: Personalized Cancer Treatments
Pharmacogenomics is a field of study that explores how an individual’s genetic makeup influences their response to different drugs. In the context of cancer treatment, pharmacogenomics plays a crucial role in personalizing the approach to therapy.
By analyzing a patient’s genetic profile, healthcare providers can determine the most effective medications and dosages, as well as identify potential adverse reactions. This tailored approach to treatment can significantly improve patient outcomes and minimize unnecessary side effects.
One of the key aspects of pharmacogenomics in cancer therapy is the identification of genetic biomarkers that can predict a patient’s response to specific drugs. This allows oncologists to select the most appropriate treatment options based on the individual’s genetic predisposition, leading to more targeted and effective therapies.
In addition, pharmacogenomics also enables the development of novel drugs that are designed to target specific genetic mutations associated with cancer. This approach, known as precision medicine, has the potential to revolutionize cancer treatment by delivering highly personalized and effective therapies.
Targeted Therapy: Exploiting Genetic Abnormalities
Targeted therapy in cancer treatment involves the use of specific drugs or other substances to attack cancer cells while doing little damage to normal cells. This approach has gained popularity in recent years due to its potential to exploit genetic abnormalities that are unique to cancer cells. By identifying these genetic changes, scientists have been able to develop drugs that specifically target and disrupt the processes that allow cancer cells to grow and spread.
One of the key advantages of targeted therapy is its potential to minimize the side effects often associated with traditional chemotherapy or radiation therapy. Rather than indiscriminately targeting all rapidly dividing cells, targeted therapy aims to hone in on the specific molecular alterations that drive cancer development. This can lead to more effective treatment outcomes while reducing the risk of harm to healthy tissues.
There are several types of targeted therapy, including monoclonal antibodies, small molecule drugs, and immune checkpoint inhibitors. Each of these approaches works in different ways to interfere with the function of specific cellular pathways that are crucial for cancer cell survival. By understanding the genetic abnormalities present in a particular tumor, oncologists can determine which targeted therapy approach is likely to be the most effective for a given patient.
As our knowledge of cancer genetics continues to expand, the potential for targeted therapy to revolutionize cancer treatment is becoming increasingly apparent. With ongoing research and development, it is likely that we will see even more precise and tailored targeted therapy options in the future, leading to improved outcomes for cancer patients.
Genetic Testing: Predicting Cancer Susceptibility
Genetic testing is a powerful tool that has revolutionized the way we approach cancer susceptibility. By analyzing an individual’s genetic makeup, scientists and healthcare professionals can identify specific genetic mutations that may increase an individual’s risk of developing certain types of cancer.
This type of testing can provide valuable information to help individuals make more informed decisions about their health, such as taking proactive measures to reduce their cancer risk or undergoing more frequent screenings for early detection.
Genetic testing can also be crucial for individuals with a family history of cancer and can help identify potential hereditary cancer syndromes, allowing for targeted interventions and personalized treatment plans.
Overall, genetic testing for predicting cancer susceptibility holds immense promise for improving cancer prevention, early detection, and personalized treatment strategies.
Gene Editing: Revolutionizing Cancer Treatment?
Gene editing is a cutting-edge technology that has the potential to revolutionize cancer treatment. By using CRISPR and other gene-editing tools, scientists can modify the genetic code of cancer cells to target and eliminate the disease. This precision approach holds promise for developing highly effective and personalized cancer therapies.
One of the most significant advantages of gene editing in cancer treatment is its ability to target specific genetic abnormalities within tumor cells. This means that treatment can be tailored to the individual’s unique genetic makeup, leading to more effective and less toxic therapies. In addition, gene editing offers the potential to correct genetic mutations that drive cancer development, potentially leading to a cure for the disease.
Furthermore, gene editing can be used to enhance the body’s immune system to better recognize and destroy cancer cells. By modifying the genetic code of immune cells, scientists can create powerful cancer-fighting agents that have the potential to eradicate the disease. This approach, known as immunotherapy, has already shown promising results in early clinical trials.
In conclusion, gene editing is poised to transform the field of cancer treatment. With its ability to precisely target and modify the genetic code of cancer cells, this technology has the potential to create highly effective, personalized, and potentially curative therapies. As research in this area continues to advance, t